Pop in fresh batteries and your remote usually works right away. That speed matters because the device does not wait for atoms to “swap places” inside the metal. Instead, a controlled chemical reaction drives electrons to move through a wire.
In plain terms, electricity is the flow of electrons. However, electrons do not travel on their own through random goo. They move when a reaction creates an electron push, then a circuit gives electrons a path.
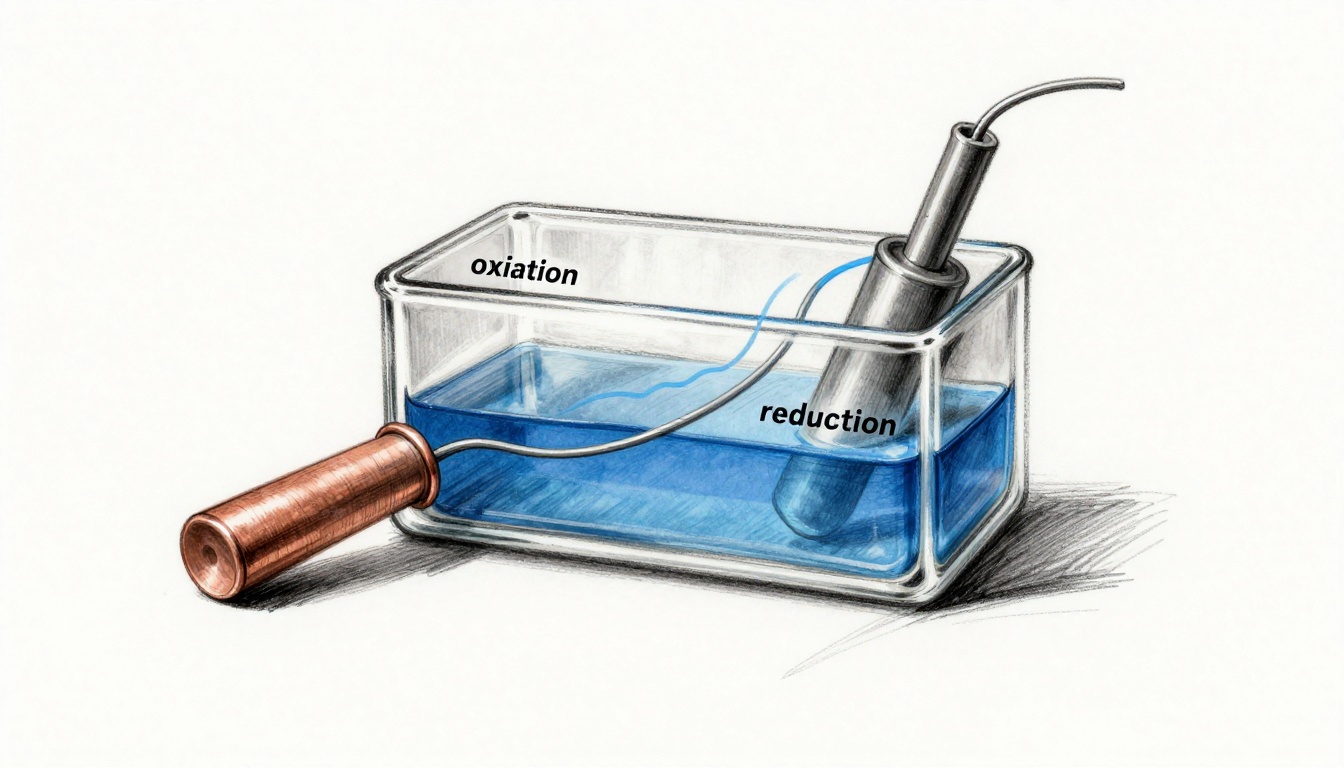
To make the mechanism predictable, real devices separate two half-reactions. One side releases electrons (oxidation). The other side accepts them (reduction). Next, the design forces those electrons to take an external route, so you can collect electrical work.
This article explains the process in three layers: redox reactions, galvanic cells, and the main battery types that show up in daily life. The goal is simple, explainable science, no PhD required.
Redox Reactions: The Secret Sauce Behind Electron Flow
A redox reaction is the coordinated set of changes where electrons transfer between substances. In this process, one reactant loses electrons. That step is called oxidation. Another reactant gains electrons. That step is called reduction. These two steps do not occur in isolation inside a functioning power source. They must balance.
A usable analogy is a “hot potato” exchange. One side has extra electrons to give. Another side has a shortage and wants them. Still, electrons cannot jump through your circuit components unless you provide a path and a separation mechanism.
A common example is the Daniell cell, which pairs zinc with copper ions. In the oxidation half-reaction, zinc metal forms zinc ions and releases electrons. In the reduction half-reaction, copper ions gain electrons and form copper metal. These changes create a voltage because the system ends up with a different chemical makeup on each side.
For clarity, the Daniell cell is often shown as zinc in a zinc-sulfate solution paired with copper in a copper-sulfate solution, with a salt bridge connecting them. A clear walkthrough of the chemistry appears in Explanation of Daniell Cell in Chemistry – BYJU’S.
Most importantly, the separation prevents electrons from taking the easy route. Without separation, electrons could recombine through direct contact and produce heat, not useful current. With separation, electrons build up and then move through a wire, which is exactly what a device needs.
Finally, the reaction must conserve charge. That requirement means electron counts must match across both half-reactions. If one side releases two electrons per event, the other side must accept two electrons per event, or the reaction stops due to charge buildup.
Oxidation: Where Electrons Get Released
The oxidation half-reaction typically happens at the anode. In the Daniell cell case, the anode uses zinc metal. During oxidation, zinc atoms leave the metal and enter the solution as ions.
A simple way to picture this is controlled “metal loss.” The zinc surface effectively dissolves into ions. At the same time, electrons remain behind in the anode material. That electron buildup raises the electrical pressure, so electrons can move outward.
The energy behind this behavior comes from differences in chemical bonding and electron “preference.” When zinc changes from solid atoms to ions, it forms new bonds with the surrounding ions in the electrolyte. Those chemical changes release energy that the cell can convert into electrical work.
If the anode and cathode were placed together in direct contact, the electrons would not travel far. They would recombine quickly. Therefore, oxidation becomes useful only when electrons have a route to leave the anode and reach a region that can accept them.
As a safety note for conceptual accuracy: “oxidation” does not automatically mean “rust” in every case. Rust is a common oxidation in air, but oxidation in electrochemistry simply means electrons are released.
For classroom-aligned reaction writing, it also helps to treat redox as half-reactions. That approach supports balancing and charge tracking. One example of this method appears in an instructional lab handout from Experiment 9 Electrochemistry I – Galvanic Cell – UCCS Chemistry.
Reduction: Electrons Find a New Home
The reduction half-reaction typically happens at the cathode. In the Daniell cell, the cathode involves copper ions in solution. During reduction, copper ions gain electrons and then deposit as copper metal.
Here, electrons arrive via the external circuit. They do not need to travel through the electrolyte. Instead, the cathode provides a surface where incoming electrons can reduce ions. Once copper ions have gained enough electrons, they form neutral copper atoms and settle out as solid copper.
This part is often misunderstood. It can look like ions “pull” electrons across the electrolyte. However, the electrolyte’s job is different. The electrolyte mainly allows ions to move so charge remains balanced. Electrons require a conductor, so the circuit provides a wire path.
As reduction proceeds, the cathode gains solid metal mass and the solution composition changes. Also, the cathode becomes more positive relative to the anode in many common designs. That polarity aligns with the direction of current in the external circuit.
Because both half-reactions run together, the system remains close to a steady operating condition. The chemical supply changes over time, and once reactants run out, the electrical output drops. Therefore, the practical “life” of a battery is tied to how long its redox pairs can sustain the electron transfer.
Galvanic Cells: Building the Device That Captures Chemical Power
A galvanic (also called voltaic) cell converts a spontaneous redox reaction into electrical energy. This conversion requires a structural arrangement that separates oxidation and reduction while still allowing charge balance.
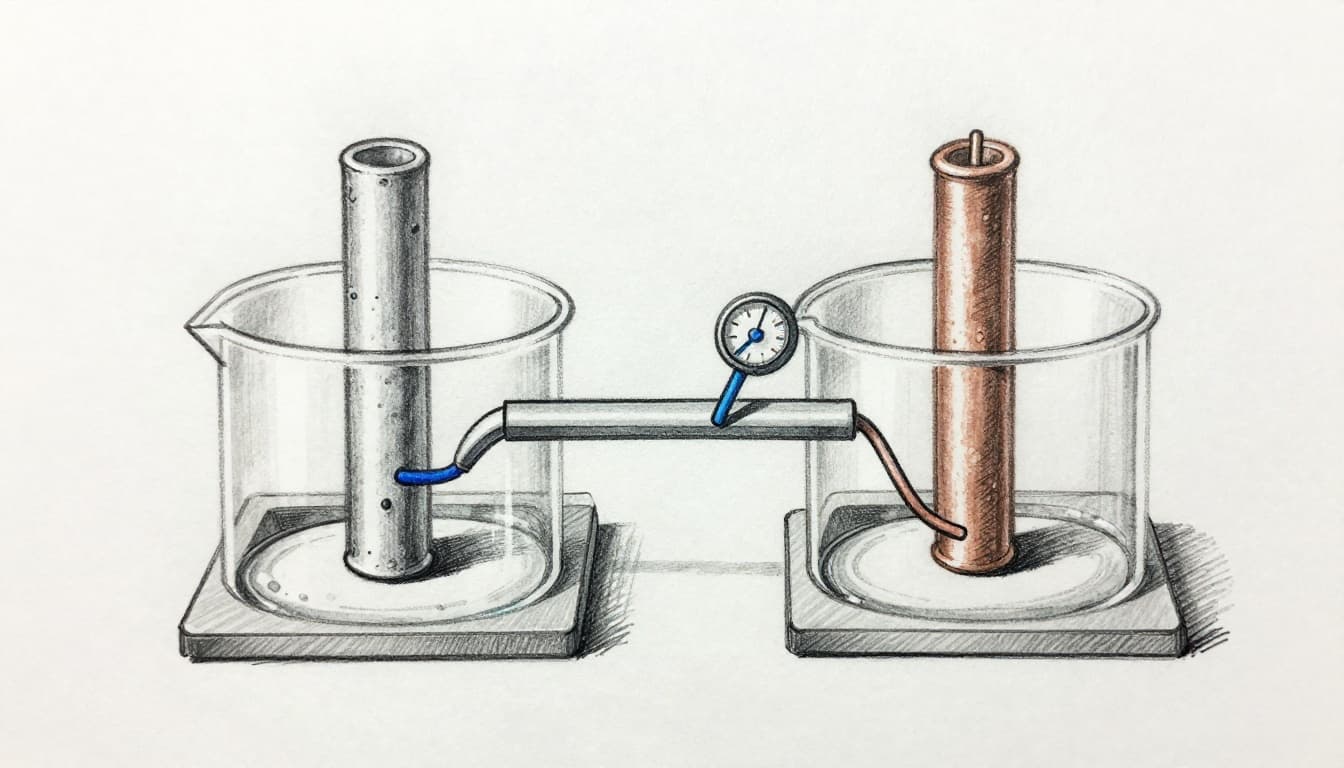
A typical setup uses two electrodes in two electrolyte regions. The electrodes are dissimilar metals (or at least different redox-active surfaces). The electrolytes contain ions that support each half-reaction. Next, the two electrolyte regions connect through a salt bridge or porous barrier. That component prevents bulk mixing, but it still allows ion flow.
The operational rule is straightforward: electrons cannot pass through the electrolyte. Electrolytes move ions, not electrons. As a result, electrons must travel through the external circuit from the anode to the cathode. A voltmeter in that external circuit measures the voltage created by the chemical separation.
Meanwhile, ions inside the salt bridge move to keep the system electrically neutral. If positive charge builds up on one side, ions move to compensate. That ion movement enables the redox reactions to continue rather than stopping from charge imbalance.
In a Daniell cell, the electron path and ion path split into two routes. Electrons take the wire route. Ions take the electrolyte and bridge route. The voltage reflects the difference between the reduction tendencies of the two half-reactions.
For a more formal description of components and cell notation, see 17.2: Galvanic Cells – Chemistry LibreTexts. That resource also describes why oxidation-reduction reactions are separated into half-reactions for balancing and analysis.
A practical expectation follows. The galvanic cell keeps producing current as long as reactants remain available and the circuit remains closed. When the circuit opens, current stops because electrons no longer move externally. Still, ion migration inside continues briefly, then the reaction rate slows as equilibrium approaches.
The Role of Electrodes and Electrolytes
Electrodes act as the places where electron transfer can occur. Therefore, the anode is the electrode where oxidation produces electrons. The cathode is the electrode where reduction consumes electrons.
Electrolytes act as the medium for ion motion. They help carry charge, so the chemical process does not immediately freeze due to imbalance. However, the electrolyte does not provide an electron highway. It cannot substitute for a metal wire in a functioning circuit.
Salt bridges or porous separators reduce the risk of direct chemical mixing. Without a separator, products formed at one side could react with reactants at the other side. That mixing would reduce the efficiency of electrical conversion and lower the available voltage.
In many common designs, the anode side runs at a negative potential relative to the cathode. Consequently, electrons in the external wire flow from anode to cathode. The polarity convention matters, especially when users connect batteries to devices with known wiring rules.
If you need one operational takeaway: electrodes determine where electrons react, and electrolytes determine how ions keep the circuit charge-balanced. Together, they define both the voltage and the rate at which the cell can deliver current.
Real Batteries Powering Your Life Today
Real batteries are designed galvanic systems. They store chemical reactants in solid or semi-solid components, then provide power when internal redox processes force electrons through an external circuit.
Battery types differ in chemistries, recharge behavior, and how they manage ion movement. Still, the same core rule holds: electrons move through wires, while ions move through internal pathways.
Here is a compact comparison of common battery families and their dominant behavior.
| Battery type | Typical use | Main operating idea | Rechargeable? |
|---|---|---|---|
| Alkaline (AA, AAA) | Remotes, flashlights | Zinc oxidation with manganese-based reduction | Usually no |
| Lead-acid (cars) | Starting and backup | Lead and lead-oxide reactions in sulfuric acid | Yes |
| Lithium-ion (phones, EVs) | Portable tech, electric vehicles | Lithium ions shuttle between electrodes | Yes |
| Fuel cells (hydrogen, others) | Vehicles, backup power | Continuous redox while fuel is supplied | Depends on system |
For an overview that connects batteries to fuel cells, consult 17.5: Batteries and Fuel Cells – Chemistry LibreTexts. That page also clarifies how a fuel cell differs from a “stored” battery.
Next, battery behavior is not only chemistry. It also depends on design choices, such as electrode surface area, electrolyte properties, and how reactants degrade under use.

Lead-Acid and Alkaline: Reliable Classics
Alkaline batteries, such as AA and AAA, use a chemistry built around zinc and a manganese dioxide-based cathode. Over time, oxidation occurs at the zinc side, which releases electrons. At the cathode, the reaction consumes electrons and forms new compounds. The internal design also supports ion movement so charge stays balanced.
From a user standpoint, the two key strengths are shelf life and steady output for low drain devices like remotes. The two key limits are energy density and recharge behavior. Most alkaline designs are not meant for repeated cycles, and capacity falls as reactants get consumed.
Lead-acid batteries, common in cars, use lead-based plates immersed in sulfuric acid. During discharge, lead at one plate oxidizes, and lead-oxide forms at the other side through reduction. These cells can deliver high current, which helps with engine starting.
However, lead-acid batteries also suffer from aging effects under repeated deep discharge. Still, they remain practical because they tolerate rough conditions and offer low cost per watt-hour for many applications.
One way to interpret both technologies is to treat them as different implementations of the same controlled electron routing. The anode and cathode do not change their purpose. Only the material systems and ion paths change.
If the question becomes “why so many chemistries exist,” a general list can help ground the idea. See List of battery types – Wikipedia for a broad map of common categories.
Lithium-Ion: Compact Power for Tech and EVs
Lithium-ion batteries also rely on redox, but the internal story differs from metal-on-metal plating in simple cells. In many lithium-ion designs, lithium ions move between electrode materials during charge and discharge. Meanwhile, electrons travel through the external circuit as the ions shift roles inside the battery.
Because lithium-ion cells are designed for high capacity, they can provide strong power output in a smaller package. That matters for phones, laptops, and electric vehicles, where weight and volume directly impact performance.
During discharge, the cathode and anode each host reactions that correspond to reduction and oxidation. During charging, the device forces those reactions to reverse direction, so the chemical reactant positions can be restored.
In operational terms, lithium-ion batteries can cycle many times, assuming the control electronics manage voltage and current within safe limits. Therefore, the chemistry and the battery management system act together. One limits stress. The other enables stable charging.
As of March 2026, solid-state designs and faster-charging chemistries have moved closer to production. Real-world announcements focus on higher energy storage per weight and improved charging times, which tie back to how electrodes and electrolytes are engineered.
Fuel Cells: Endless Power with Fuel Supply
Fuel cells produce electricity through redox reactions as long as fuel and oxidant are supplied. In a common hydrogen-oxygen fuel cell, hydrogen is oxidized at one electrode, and oxygen is reduced at the other.
Unlike a battery, a fuel cell does not rely on a fixed amount of chemical reactant stored inside the unit. Instead, it draws reactants continuously. Therefore, the power output can remain steady over time, limited mainly by fuel supply and system conditions.
This feature supports applications where long runtimes matter, including vehicles and certain backup systems. It also helps explain why fuel cells attract interest alongside better battery designs.
However, fuel cells still obey the same core constraint: electrons do not move through the electrolyte. They go through the external circuit, then do useful work. The electrolyte and separator manage ion flow and charge balance, so the electrochemical process can keep going.
From a systems view, fuel cells and batteries often complement each other. Batteries store energy for bursts and steady use. Fuel cells can provide longer runtime when fuel is available.
Why Chemical Electricity Shapes Our Energy Future
Chemical reactions produce electricity through a repeatable framework: redox separates charge, galvanic designs route electrons through wires, and battery construction packages those reactions into practical devices. Because the conversion is tied to materials, improving performance means improving materials.
That means better electrodes, better electrolytes, and better ways to manage reaction rates without excessive wear. In turn, these upgrades translate to higher energy density, safer operation, and longer service life.
In energy planning, electricity from chemical systems also enables portability. A phone cannot carry a power plant, but it can carry a compact electrochemical setup. Likewise, electric vehicles must store energy onboard, so chemical-to-electrical conversion defines range and driving comfort.
At the policy and infrastructure level, chemical electricity allows more fuel options. Some energy sources can be converted into electricity through electrochemical pathways, including fuels that can be produced from renewable inputs. That property can reduce dependence on a single supply chain, if production scales responsibly.
Meanwhile, market progress keeps accelerating. Solid-state battery announcements and faster charging timelines in 2026 signal that better electron and ion management is translating into measurable outcomes. Even incremental improvements can shift how people use electric transport.
The mechanism behind it remains the same as the first remote battery. Redox forces electrons through a wire, and controlled chemistry turns that movement into usable power.
Conclusion
Chemical reactions produce electricity when redox reactions create an electron push, then a galvanic design routes those electrons through a circuit. Oxidation releases electrons at the anode, reduction accepts them at the cathode, and the electrolyte and separator manage ion flow so the process stays stable.
Every battery you use works on this same structure, whether it is alkaline, lead-acid, lithium-ion, or a fuel cell. In practice, you see it in flashes, engine starts, and charging cycles, because electrons keep taking the external wire path.
Try a simple experiment, such as a lemon battery with two different metals. If a small cell can power a light, the concept becomes concrete. When you pop in batteries again, the science behind that moment is still the same controlled electron flow.