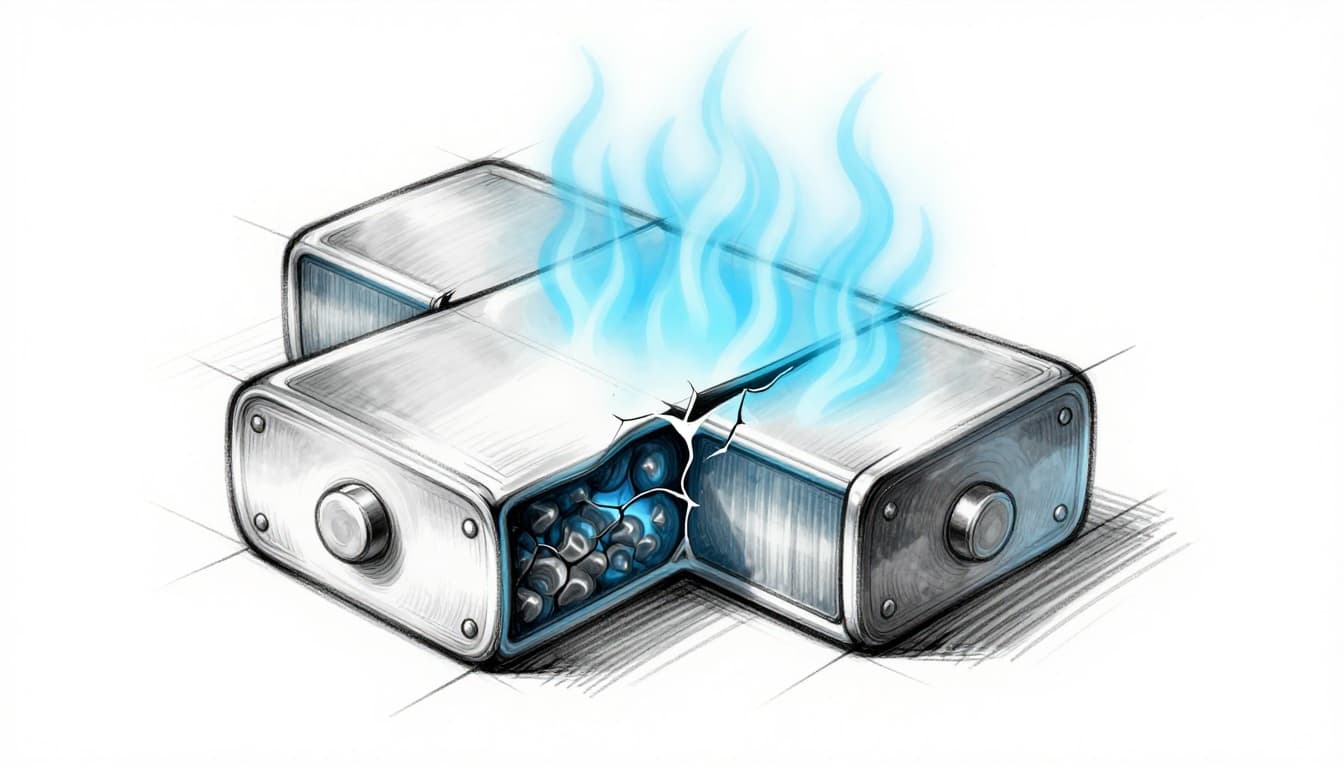
A battery does not fail all at once, unless something in its cells breaks. More often, you see gradual battery degradation first, with capacity shrinking and runtime falling. Then, under the right stress, battery failure can occur, including sudden shutdowns or rare fire events.
Heat, cold, and charging habits drive most of the wear. Chemical side effects build up until the cell can no longer deliver power. In addition, manufacturing defects and physical damage can create fast paths to failure.
So, what causes battery failure and degradation in day-to-day phone and EV use? The answer matters because you can reduce risk by changing a few behaviors, supported by 2026 EV and smartphone safety trends.
Why Heat and Cold Are Battery Life Thieves
Battery chemistry runs on time and temperature. When temperature rises, internal reactions accelerate. When temperature drops, the battery cannot move ions as easily. As a result, both extremes push cells toward permanent damage.
For heat, the problem is not only external temperature. The cell can get hotter during charging, fast charging, climbing hills, or even hot weather sitting in a vehicle. After that, the battery may form thicker protective film on electrodes. This film is called the SEI layer, and extra growth blocks charge flow. Over time, power drops and the cell becomes less stable.
For cold, the key risk is higher internal resistance. In practice, that means you get less usable power. You also increase the chance that the battery will act unevenly across cells in a pack. Meanwhile, repeated cold starts can add stress that later shows up as faster loss.
The following effects are typical under temperature extremes:
- High heat: SEI thickens faster, parts inside can degrade, and resistance can rise.
- Cold weather: output falls, charging gets less efficient, and lithium plating risk rises in some conditions.
- Temperature swings: rapid changes can increase mechanical stress inside the cell.
- Hot charging: charging adds heat, and heat accelerates wear during the same session.
Ever notice your phone battery draining faster on hot days? That is consistent with battery behavior under elevated temperature. Also, modern 2026 EVs continue to refine active cooling, because temperature still drives failure risk and lifetime cost.
A peer-reviewed mechanism overview describes how high temperature aging changes lithium-ion battery components and accelerates degradation pathways, especially during prolonged hot storage or operation (see high-temperature aging mechanism).
High Heat Sparks Dangerous Chemical Changes
When heat rises above normal operating conditions, side reactions speed up. The battery starts to create more SEI material than it should. Because the SEI layer grows, more lithium becomes trapped and less is available for the main reaction.
In practical terms, that means capacity fades. It also means the cell can run hotter under load because internal resistance tends to increase. On a bad day, this combination can contribute to thermal runaway risk, where heat feeds more heat.
Heat also affects the materials that bind electrodes and hold electrolyte in place. As these parts degrade, the cell may lose structure and efficiency. Later, you may observe rapid capacity loss, higher voltage sag under load, and reduced peak power.
The same general failure logic applies to phones and EVs, even if designs differ. For EVs, battery packs often include sensors and thermal management, but hot climates and repeated heavy charging can still overwhelm margins. In other words, the battery does not need abuse every day. It needs the right stress pattern often enough.
Cold Weather Stresses Batteries from the Inside
Cold does not “burn” batteries like heat does. Instead, cold limits performance and increases stress during charging. In low temperatures, ions move more slowly. As a result, the battery may show slower response, higher internal resistance, and weaker output under load.
Charging in cold conditions can become a risk control issue. If the battery cannot accept charge efficiently, lithium may deposit in the wrong places. That is the start of lasting damage, because deposited lithium can later affect cycling stability.
You can use a simple analogy. Cold conditions act like a frozen pipe. Flow becomes harder, even if you still try to force it. When you later warm the system, some of the damage has already formed.
Storage behavior adds another layer. Temperature and state of charge jointly shape long-term degradation. A modeling study on storage degradation shows that temperature and the battery’s state of charge can change how fast capacity fades over time (see temperature and state-of-charge storage).

In short, cold often raises short-term resistance. Then, repeated use in cold can create long-term wear.
How Charging Cycles Wear Down Batteries Over Time
Battery wear accumulates through use. Each charge and discharge event drives chemical change. Even when the battery looks “fine,” internal aging can continue. This process is often tied to SEI growth, electrode wear, and loss of active material.
Industry and monitoring data commonly describe meaningful runtime loss after hundreds of cycles for EV packs, with capacity often falling toward 80% after roughly 500 to 1,000 cycles. Smartphones with daily use can reach similar performance declines in about one to two years, depending on heat, charge patterns, and average state of charge.
You can think of cycling as footsteps. Each step may seem small, but it still wears a path deeper. Over many sessions, the worn path becomes a problem, not just a surface change.
A key operational point follows. Batteries do not degrade only from full charge or full drain. Partial charging still counts as cycling, because ions still move and chemical side effects still occur.
The Hidden Damage from Everyday Charge-Discharge Loops
Even “normal” use can create a cycle pattern. In many daily workflows, the battery charges, then discharges, then rests at a high state of charge. That resting time can add additional aging stress, especially at elevated temperatures.
Most wear shows up as capacity fade and power limits. You may not see a sudden shutdown. Instead, you may see a shorter runtime, less peak power during camera use or gaming, and slower acceleration in EVs.
Under repeated cycling, electrode surfaces change. Micro-cracks may form, and particles can break away. Also, the SEI layer continues growing. Because SEI consumes lithium, the cell gradually has less “usable” lithium to store charge.
For practical risk control, the main failure to avoid is not one bad event. It is repeated stress that overlaps: high heat plus charging plus high state of charge.
Fast Charging and Overcharge Risks Explained
Fast charging pushes the battery harder. It can raise internal current and speed up stress processes. As a result, fast charging can increase degradation faster than slower charging. This is consistent with recent reporting on how high-power fast charging accelerates EV battery degradation, based on industry studies (see fast charging EV degradation report).
Overcharge risk also matters. If the cell voltage stays too high, the battery may experience stronger side reactions. That can increase gas formation and stress on the electrode structure.
Over-discharge can be harmful too. If the cell voltage drops too low, electrode layers and materials can degrade. Then, the battery may not recover fully.
Additionally, bad timing and control failures can worsen outcomes. If a battery management system, charger firmware, or communication does not manage limits correctly, the cell can spend longer outside safe operating regions.
The operational standard is clear: frequent extreme charging conditions increase the probability of both degradation and, in rare cases, failure events.
Chemical Enemies Lurking Inside Every Battery
Inside a lithium-ion cell, failure often starts as a microscopic problem. Then, heat and charging stress turn it into something bigger. This is why the phrase “battery failure” can be misleading. You are usually dealing with cell chemistry that has crossed thresholds.
The most important chemical risks include lithium dendrites and electrolyte breakdown. When these happen, the battery may lose efficiency first. If the issue grows, it can create an internal short, then rapid heat rise, then severe failure.
What if tiny needles are destroying your battery? That is not a metaphor. Dendrites can form under certain stress conditions and can disrupt internal safety barriers.
A review of battery failure causes and mitigation methods summarizes how failures can originate from failure mechanisms like abnormal lithium growth, degradation of materials, and separator-related issues (see battery failure cause and mitigation review).

Dendrites: Needle-Like Growths Causing Shorts
Dendrites are small metal structures that may form during charging when conditions push the cell out of balance. If growth reaches the wrong area, it may pierce the separator. When that occurs, the cell can short internally.
Shorts inside a cell are not always immediate disasters. Sometimes the battery detects rising heat and voltage changes. Then it shuts down or limits output. Other times, the heat rise can continue, creating thermal runaway risk.
Recent reporting also points to the microscopic “metal thorns” idea. In March 2026, coverage highlighted how researchers observed dendrite formation that can lead to short-circuiting behavior (see microscopic metal thorns).
Real-world examples vary by product type and manufacturing batch. Still, the mechanism is similar. Fast charge, overcharge, or uneven cell stress can increase dendrite risk.
Electrolyte Decay and Its Chain Reaction
Electrolyte is not inert. It participates in the cell’s chemistry and also forms parts of the SEI and other layers. Over time, electrolyte decomposition can increase resistance and reduce stable operation.
As resistance increases, the battery runs warmer when under load. That creates a chain reaction. Heat speeds up more reactions, which increases resistance again.
In simple terms, the battery becomes less efficient. Then, it becomes more stressed. Over time, the cell may show higher voltage drop under load, faster heating during charge, and earlier shutdowns under protection logic.
Because electrolyte decay stacks with cycling and temperature stress, it can accelerate the point where degradation becomes failure risk.
Factory Flaws and Physical Hits That Doom Batteries
Not all failure causes originate from user habits. Some start during manufacturing. Some start from physical damage. In both cases, the defect or damage provides a pathway for internal shorting or unsafe heat buildup.
Manufacturing defects may include poor separator quality, contamination, or misalignment of internal components. If internal parts do not meet tolerances, current paths can form where they should not. Then, heat can rise during charging, especially when the control system is trying to manage a cell already predisposed to failure.
Physical damage includes drops, crushes, or impacts. These events can deform the housing and stress internal components. Even small impacts can crack internal structures over repeated vibration and use.
It is also acceptable to treat these as separate categories. Chemical degradation is an internal aging process. Physical defects add a direct risk path, especially if the separator or internal wiring is compromised.
Common Defects Straight from the Production Line
When a manufacturing flaw exists, the risk may appear in specific batches. Reports of EV safety actions in 2026 show that some battery fire risks trace back to battery issues that involve internal alignment or module behavior during operation.
For example, Reuters reported that Volvo issued a recall involving battery fire risk in certain EV models, tied to specific concerns about battery pack overheating (see Volvo recall over battery fire risk).
These events do not mean “all EVs are unsafe.” Instead, they show that even controlled production can still produce defects that require field action. Also, once a defect exists, temperature and charging patterns can trigger the problem sooner.
Drops, Crushes, and Everyday Abuse
Physical damage can be faster than chemical wear. A drop can shift internal layers or damage the separator. A crush event can deform the cell can and create stress points. Vibration also matters, especially for e-bikes, off-road use, and vehicles with harsh ride conditions.
In phones, battery failure from physical damage often shows up as swelling, rapid heat, or sudden shutoffs. In EV packs, internal wire damage and module deformation can also contribute to abnormal heating.
The main administrative takeaway is simple. Users control the environment more than the chemistry. That means you should treat physical impacts as potential battery risk events, not minor cosmetic issues.
If you observe bulging, repeated overheating, or unusual smells, you should stop using the device and follow manufacturer or safety guidance. A battery with a damaged internal structure can fail unpredictably.
Conclusion: Temperature, Cycles, Chemistry, and Defects Set the Outcome
Across phones and EVs, the most common drivers of what causes battery failure and degradation are temperature extremes, repeated cycling, chemical side reactions, and manufacturing or physical damage.
If one factor dominates your decision-making, it should be temperature. Heat accelerates internal changes. Cold raises stress during charging and output. Meanwhile, frequent fast charging and high state-of-charge habits shorten the usable life of many cells.
In 2026, safety systems and thermal controls are better, but they do not remove all risk. Practical next steps remain consistent: avoid high heat exposure, reduce frequent extreme charging, and treat impacts as a potential battery safety issue.
Share your battery experience in the comments. If your phone or EV degraded faster than expected, describe what the conditions were.
