Your phone drops to 2% in the middle of a busy day, and you need power now, not later. In practical terms, a battery is a device that stores chemical energy and converts it into electrical energy to run everyday gadgets, like flashlights and remotes. When you understand battery basics and how batteries work, you can choose the right type for the job and follow safe use rules that reduce leaks, overheating, and early failure. For example, you can avoid the common mistake of mixing old and new batteries, which can cause devices to underperform or shut down early. Next, the first section covers the main components that make a battery function the way it does.
Break Down the Key Parts Inside Every Battery
A battery shall be treated as a controlled chemical system with three internal parts that operate together. Each part has a distinct duty, and each duty exists to keep electrons moving in one path while ions move to maintain charge balance. If one part fails, the battery output can drop, heat can rise, and in some cases damage can occur. For technical background on how common lithium battery materials are arranged, see Purdue’s ViPER overview of Li-ion components.

A simplified diagram description is as follows:
- Anode (left, negative side): releases electrons by oxidation
- Electrolyte/separator (center): allows ion movement, blocks direct electron flow
- Cathode (right, positive side): accepts electrons by reduction
Anode: The Electron Launchpad
The anode shall be assigned as the negative side, because it performs oxidation, meaning it loses electrons. In most common battery chemistries, the anode material acts as the source of those electrons. For example, zinc is a well-known anode material used in many alkaline batteries.
Operationally, the anode releases electrons into the external circuit. Those electrons then travel through the device load, providing usable electrical power. Meanwhile, chemical change continues at the anode surface until the reactions reach their limits.
Use the airport analogy for clarity: the anode operates like a busy airport sending out planes. The planes represent electrons, and they must depart in an orderly way so your device can “receive power” on schedule.
Cathode: The Electron Receiver
The cathode shall be assigned as the positive side, because it performs reduction, meaning it gains electrons. At the cathode surface, incoming electrons drive the chemical reaction that turns battery chemistry into steady electrical output during use.
In many real batteries, cathode materials include manganese oxide (often described as MnO₂) for alkaline systems. In other chemistries, the cathode material varies, but the functional requirement remains consistent: the cathode must accept electrons and complete the circuit from a chemical standpoint.
For an analogy, the cathode behaves like a parking lot for arriving planes. Electrons arrive, then the cathode reaction “hosts” them. As electrons accumulate and react, charge flow stays balanced with ion movement, and the battery continues to deliver power until active materials are consumed.
Electrolyte: The Ion Highway
The electrolyte shall function as the ion highway by allowing ions to move inside the battery while preventing direct electron contact. This separation is necessary, because electrons must not short-circuit internally. Instead, electrons should pass through the external circuit where the device draws current.
In common designs, the electrolyte can be a liquid (for example, acid-based or salt-in-solvent systems) or a gel or paste (including gel separators used in some battery forms). Regardless of physical state, the electrolyte conducts ions so charge balance can persist while reactions proceed at both electrodes.
Use the bridge analogy for clarity: ions cross the electrolyte “bridge,” while electrons take the external wire route. This design constraint forces current to go where it is intended, instead of taking an internal shortcut that degrades performance and can increase heating risk.
Step by Step: The Chemical Reactions That Create Electricity
This section sets out, in operational order, how a battery produces electricity by converting chemical changes into controlled electron flow. You can treat the battery as a regulated chemical system that “moves” charge so your device can run, like a timed feed that turns chemistry into current. For clarity, the pathway splits into two tracks: electrons move through your device, while ions move inside the battery.
What Happens in Primary Batteries
Primary batteries (non-rechargeable) operate under a one-way duty cycle, meaning the chemical reactions proceed until active materials deplete. In an alkaline AA cell (common in remotes), the cell relies on the zinc and manganese dioxide chemistry in a potassium hydroxide electrolyte. When the circuit closes, the internal reactions proceed in the direction that produces power, and the products accumulate in a way that eventually stops efficient flow. For an applied reference on alkaline manganese dioxide chemistry, see Energizer’s alkaline battery chemistry guide.
Follow the electron and ion process as a step sequence:
- Device connects the circuit: when you insert the battery and switch on the remote, the external path completes.
- Oxidation at the anode (negative side): zinc loses electrons at the anode surface. Those electrons enter the external circuit.
- Electron flow through the device: electrons move from the battery through the load, which includes the bulb filament in a simplified example.
- Reduction at the cathode (positive side): manganese dioxide gains electrons at the cathode, completing the electrochemical balance.
- Ions travel internally: ions move through the electrolyte to maintain charge balance, while the electrolyte does not route electrons directly.
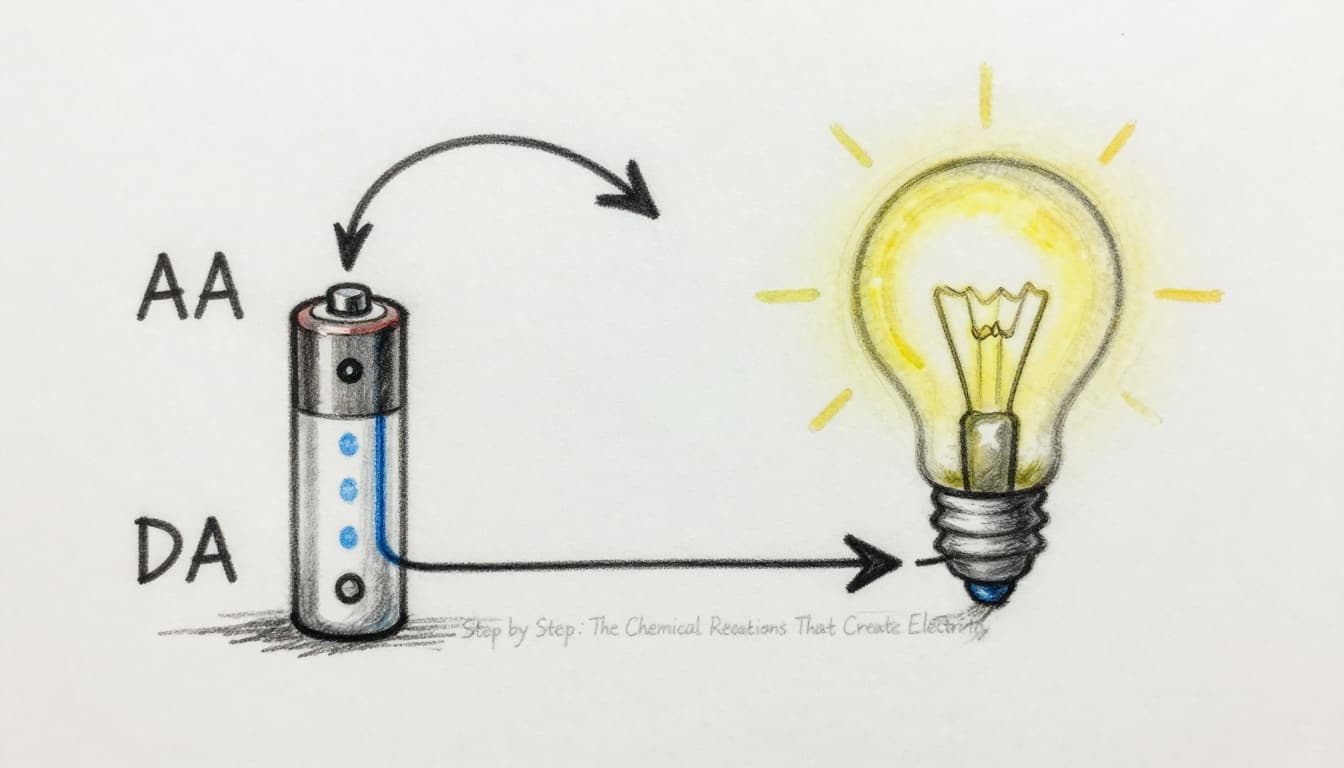
In short, the battery does not “store electrons” for later use. Instead, it runs chemical reactions that keep replenishing electrons at the anode only until the chemistry can no longer sustain the needed rate.
Rechargeable Batteries in Action
Secondary batteries (rechargeable) apply the same basic rule, electrons through the device and ions inside the battery, but charging changes the allowed reaction direction. During discharge, the cell behaves like a primary cell, producing current through oxidation at one electrode and reduction at the other. During charging, the battery controller enforces a reverse, so the internal reactions backtrack and restore the original chemical states.
For lithium-ion batteries, a common operating description is that lithium ions migrate between electrodes through the electrolyte, while electrons travel through the external circuit (through the charger during charging, and through the device during discharge). For a plain-language process overview, refer to how lithium-ion batteries work (U.S. Department of Energy).
The practical operational sequence is therefore:
- Discharge (phone running): electrons leave the anode, move through the phone load, and arrive at the cathode.
- Ion migration (inside the cell): lithium ions move through the electrolyte to maintain charge balance.
- Charging (phone plugged in): an external power source forces the reaction directions to swap.
- Reversible chemical states: the battery restores its active materials so the cycle can repeat.
The compliance requirement for safe use follows from these facts: charging requires control. If the charging conditions run outside designed limits, side reactions can occur, and the cell can degrade or heat up.
Everyday Battery Types and Where You’ll Find Them
Everyday batteries typically fall into two duty classes: primary (non-rechargeable) and secondary (rechargeable). In daily use, you select by both chemistry and job profile (low-drain vs high-drain), because a battery that fits the size may still underperform under load. For a general shopping reference that also lists common household choices, see The Home Depot’s battery types guide.
In short, alkaline and zinc-carbon serve quick, budget tasks, while lithium-ion and lead-acid cover heavier runtime needs. A practical comparison of what you will see in US homes is shown below.
| Battery type | Typical forms | Common locations | Primary use fit |
|---|---|---|---|
| Alkaline | AA, AAA, 9V | Remotes, clocks, toys | Low to medium drain |
| Zinc-carbon | AA, AAA | Basic toys, wall clocks | Lowest-cost, light duty |
| Lithium primary | Coin cells, CR series | Fobs, flashlights | Long shelf, higher current |
| NiMH (rechargeable) | AA, AAA | Rechargeable toys, cameras | Frequent recharging |
| Lead-acid | Car battery | Vehicles, some backups | High current, heavy housing |
| Lithium-ion (rechargeable) | Packs, cylinders | Phones, power tools | High-drain, compact power |

Alkalaline and Zinc-Carbon for Quick Jobs
For household quick jobs, alkaline and zinc-carbon represent the most common primary battery options. Alkaline cells usually deliver steadier output and longer life in common items, including clocks, TV remotes, and many battery-powered toys. As a result, alkaline is typically the default choice when the device manual does not require a specific chemistry.
Zinc-carbon batteries usually cost less, but the design prioritizes low power draw. Therefore, they tend to fit basic clocks, simple toys, and low-drain devices where runtime expectations are modest. If a zinc-carbon cell runs a high-drain device, voltage sag can appear early, causing weak operation or early cutoff. In practice, both types are one-use and should not remain installed after they fail.
Powerhouses Like Lithium-Ion and Lead-Acid
For high-drain devices and long runtime requirements, the main secondary options are lithium-ion and lead-acid, each built for a distinct operating environment. Lithium-ion batteries appear in phones, laptops, power tools, and many motorized toys, because their energy density allows compact packs with strong output. However, recharge control remains required, since damaged cells can overheat under fault conditions.
Lead-acid batteries appear primarily in cars and some standby backup systems, because they deliver very high starting current. Also, the chemistry accepts heavy current draw without needing the small-size format that many household products require. The tradeoff is weight and limited flexibility, so lead-acid rarely fits everyday remotes or clocks.
A Peek at Battery History, Safety Rules, Myths, and What’s Next
Battery history reads like a chain of “permission slips” that made electricity portable. Each step solved a specific problem, then created a new one to fix. As a result, today’s batteries exist because inventors learned what works under load, what fails under stress, and what safety controls must cover.
Milestones That Changed Our World
Three milestones stand out because each one unlocked broader use of stored power.
- 1800, Volta’s voltaic pile: Alessandro Volta demonstrated a practical way to produce steady current using stacked metals (like zinc and copper) with a chemical path in between. It did not look like a modern cell, yet it established the core idea of controlled electrochemical output.
- 1859, lead-acid takes form: Gaston Planté’s lead-acid concept later supported repeatable charging and discharging, which is a key behavior behind today’s rechargeable systems. Cars and backup power owe their basic pattern to this approach.
- 1991, lithium-ion goes mainstream: Modern lithium-ion designs delivered high energy density and broad consumer adoption, enabling phones, laptops, and compact power packs.
For a fuller timeline, see battery evolution from pile to lithium and battery history at BatteryGuy.

Smart Safety Habits and Myth Busters
Battery safety is not optional paperwork. It is a set of practical controls that reduce leakage, overheating, and short circuits. Because battery chemistry stores energy, misuse can turn that stored energy into heat.
Use these smart, everyday habits:
- Do not mix brands or ages in the same device. Use matched cells so the output stays consistent.
- Keep terminals clean and dry. If corrosion appears, replace the battery rather than “fixing” it inside the device.
- Avoid heat and direct sun. If a battery case feels hot, stop using the device.
- Use the correct charger for rechargeable packs. An incorrect charger can push voltage or current beyond design limits.
- Store damaged batteries separately and out of reach of children. When a pack looks swollen, do not continue testing it.
Common myths should be treated as safety risks, because they often lead to the wrong action:
- Myth: Batteries always “self-discharge,” so freezing helps.
Truth: Freezing can help slow aging for some chemistries, but it does not make a damaged cell safe. In addition, condensation risk exists when you remove a cold pack. - Myth: All batteries behave the same in cold weather.
Truth: Types differ. Some deliver weaker output in cold, while others manage it better through design and chemistry.
For a basic safety reference, follow Battery Safety Tips from ESFI, and for public-handling risks, reference Battery University safety in public.
Hot New Battery Tech on the Horizon
The “next” battery work is not limited to higher numbers. It also targets fewer failures, faster charging, and better waste control, especially for EVs and grid storage (solar and wind). In practical terms, these advances aim to make batteries act more like reliable infrastructure and less like delicate components.
Right now, three technology directions receive the most attention:
- Solid-state batteries: These use a solid electrolyte instead of flammable liquid forms. As a result, they can improve safety and may allow faster charging and higher energy density. Recent reports indicate vehicle trials and pilot production are targeting around 2027, with Toyota pointing to prototypes in that window.
- Sodium-ion batteries: These focus on lower cost materials (sodium instead of lithium) and can support good performance in some conditions, including cold. Their main tradeoff is energy density, but scale and price may help adoption.
- Recycling advances: The industry direction emphasizes higher recovery rates and better sorting methods, so old packs return as feedstock rather than waste. For EVs and renewables, improved recycling matters because it reduces raw material strain.
As of March 2026, the practical takeaway is clear: solid-state progress remains the headline, while sodium-ion and recycling improvements work in parallel. The next generation will likely arrive as a sequence of pilot fleets, then wider rollouts once safety and durability data match the promise.
Conclusion
A battery is a battery definition you can apply to everyday use: it stores chemical energy and converts it into electrical energy, so devices can run when you switch them on. Because the anode, cathode, and electrolyte each play a set role, the battery output follows a clear process during discharge (and charging, when the design is rechargeable).
For correct battery basics, focus on battery types and fit. Primary options work until active materials deplete, while rechargeable batteries use controlled chemistry changes to repeat the cycle. Battery safety follows the same rule set, so you should not mix ages or brands, keep terminals clean and dry, and stop use if heat or damage appears.
Next step, check the batteries already in your home devices (date, type, and condition), then recycle used packs through local drop-off rules. What’s your go-to battery-powered gadget when you need it most?
